Sodium Bisulfate: Formula, Density & More
Understanding Sodium Bisulfate and Its Characteristics
Sodium bisulfate is a dry, granular acid salt formed through the reaction of sulfuric acid with sodium chloride in a continuous flow reactor.
Sodium Bisulfate Formula
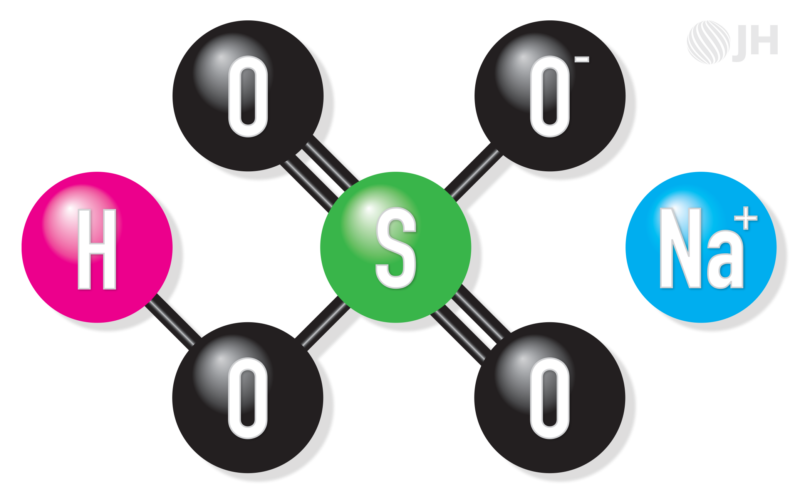
The chemical formula of sodium bisulfate is NaHSO4. It dehydrates at 136 °F (58 °C) and once cooled again it is freshly hygroscopic.
Sodium bisulfate, which occurs as white crystals or granules that are soluble in water, functions as an acidifier. The product is non-hazardous to ship and store.
Sodium Bisulfate Chemical Properties
| Molar mass | 120.06 g/mol (anhydrous) 138.07 g/mol (monohydrate) |
| Density | 2.742 g/cm3 (anhydrous) 1.8 g/cm3 (monohydrate) |
| Melting point | 315 °C (anhydrous) 58.5 °C (monohydrate) |
| Solubility in water | 28.5 g/100 mL at 25 °C 100 g/100 mL at 100 °C |
| pKa | 1.99 |
| CAS# | 7681-38-1 (anhydrous) 10034-88-5 (monohydrate) |
Also known as sodium hydrogen sulfate, sodium bisulfate is highly water soluble. It is safe and stable in dry conditions but dissociates readily in water. Unlike a neutral salt, sodium bisulfate is an acid salt because upon dissociation, a H+ ion is released. As a result, sodium bisulfate will react with bases and oxidizing agents.
Read more about sodium bisulfate hazards.
Sodium Bisulfate Molar Mass
Molar mass is defined as the sum of the atomic weights of each element in the compound.
Broken down, the sodium bisulfate molar mass can be shown as:
1
sodium atom
1
hydrogen atom
4
oxygen atom
1
sulfur atom
The atomic weights and the weight percent of each element in sodium bisulfate is as follows:
| Element | Symbol | Atomic Weight | # of Atoms | Weight Percent |
|---|---|---|---|---|
| Sodium | Na | 22.9 | 1 | 19.1% |
| Hydrogen | H | 1.0 | 1 | 0.8% |
| Oxygen | O | 15.9 | 4 | 53.3% |
| Sulfur | S | 32.0 | 1 | 26.7% |
Sodium Bisulfate Uses
Due to its stability and chemical makeup of elements found naturally in the environment, sodium bisulfate is used in a wide variety of applications, including:
OHIO
30354 Tracy Road Walbridge, Ohio 43465
SOUTH CAROLINA
4433 Richburg Road Richburg, South Carolina 29729
(888) 858-4425
